|
Hydrogen molecules are first broken up into hydrogen atoms (hence the atomic hydrogen emission spectrum) and electrons are then promoted into higher energy levels. The high voltage in a discharge tube provides that energy. But if energy is supplied to the atom, the electron is excited into a higher energy level, or even removed from the atom altogether. When unexcited, hydrogen's electron is in the first energy level-the level closest to the nucleus. These images show (a) hydrogen gas, which is atomized to hydrogen atoms in the discharge tube (b) neon and (c) mercury. Figure 1.4.3 : The Emission Spectra of Elements Compared with Hydrogen. The line spectra of several elements are shown in Figure 1.4.3. The wavelengths of lines in hydrogen’s spectrum will be measured by reflecting the light from the spectroscope’s diffraction grating onto a ruled. These so called line spectra are characteristic of the atomic composition of the gas. Why does hydrogen emit light when excited by a high voltage and what is the significance of those whole numbers? Hydrogen’s emission spectrum will be observed by applying a high-voltage discharge to a tube filled with hydrogen gas and viewing the light emitted by the excited atoms with a spectroscope. Hydrogens lines in the region of purple light seem to match up, but the lines around 485 nm and 655 nm just barely do not. It also looks at how the spectrum can be used to find the ionisation energy of hydrogen. Each line can be calculated from a combination of simple whole numbers. THE ATOMIC HYDROGEN EMISSION SPECTRUM This page introduces the atomic hydrogen emission spectrum, showing how it arises from electron movements between energy levels within the atom. The lines in the hydrogen emission spectrum form regular patterns and can be represented by a (relatively) simple equation. H-he-hg Emission Spectra by Science Photo Library, Hydrogen Emission Spectrum, Hydrogen spectrum spectra absorption: wavelength frequency electromagnetic. I doubt there is enough hydrogen gas in a discharge tube to do this, since by design they are meant to display clean line spectra. The planetary model for the hydrogen atom that explained the emission spectrum of the hydrogen gas was proposed by Bohr, who made the following assumptions. The origin of the hydrogen emission spectrum If you then ramp up the density of the gas, the recombination continuum would get stronger, gradually filling in the gaps between the emission lines until you approached a blackbody spectrum. 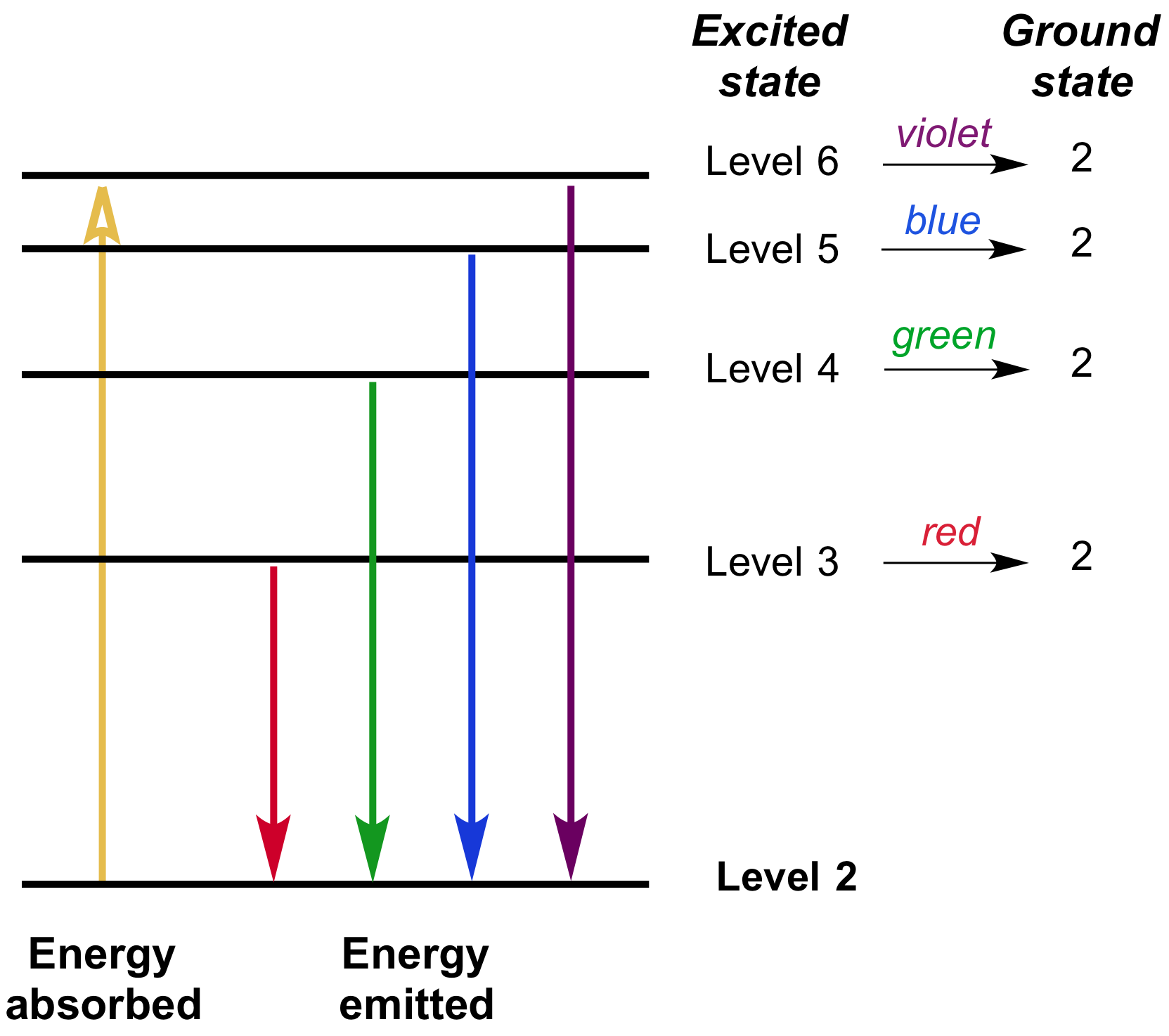
The various combinations of numbers that can be substituted into this formula allow the calculation the wavelength of any of the lines in the hydrogen emission spectrum there is close agreement between the wavelengths generated by this formula and those observed in a real spectrum.Ī modified version of the Rydberg equation can be used to calculate the frequency of each of the lines: The model bears his name because of his interpretation of the emission spectrum of hydrogen: If a small amount of hydrogen gas is confined within a glass tube. 
moderate - 660 nm Red spectral line (1 pts) General observations about the lamp and spectrum: Saved Normal BIU Xl X- ELE fx. In other words, if \(n_1\) is, say, 2 then \(n_2\) can be any whole number between 3 and infinity. Table view List view Table 1: Hydrogen gas emission spectrum Wavelength 410 nm Intensity faint Violet spectral line Choose faint Blue-violet spectral line 435 nm Choose moderate 485 nm Blue-green spectral line Choose. \(n_1\) and \(n_2\) are integers (whole numbers).Explain the connection between the emission spectrum of hydrogen gas and the Bohr model of the atom. Figure 1.4. 
Calculate the wavelenths colors and frequencies of the emission spectrum of hydrogen gas. These so called line spectra are characteristic of the atomic composition of the gas. \): When light from a hydrogen gas discharge tube is passed through a prism, the light is split into four visible lines.\right)\] Identify the emission spectrum of hydrogen gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
